Early season root development strongly influences crop season performance. A key factor for optimum root growth is phosphorus nutrition. Phosphorus is the energy molecule of the plant. Root growth, especially early season, occurs under naturally adverse conditions in the soil and this stress point demands abundant energy.
The plant available form of phosphorus is PO4=. The strong and reactive negative charge of phosphorus creates chemical challenges in the soil environment. Positively charged elements, such as calcium, react with phosphorus rendering the phosphorus unavailable for plant assimilation. The vast majority of soil reserve phosphorus is chemically unavailable or “tied-up”, meaning it is reacted in compounds that the plant cannot readily use.
Conventional phosphorus sources such as phosphoric acid, or its derivative 10-34-0, contain 100% plant available phosphorus when they arrive at the ranch in their concentrate form. This however changes quickly once they are mixed with water or applied to the soil as the phosphorus ties-up with calcium, zinc, and other cations. In order to make phosphorus more available, several tactics and technologies have been developed to improve plant availability. We will discuss three: soil chemistry, soil biology, and complexing.
The adverse soil chemical reactions that create plant unavailable compounds can be reversed by reacting these with acid forming or acid inputs. When and where this is appropriate is site specific and requires a soil analysis. Much of California’s coastal soils are high pH and acid forming inputs can be very beneficial. These include elemental sulfur and ammonium-based nitrogen sources. In many cases these have positive but short-lived efficacy improving plant phosphorus availability.
Improving biological activity in soil improves phosphorus availability in two ways. First, it creates increased carbon dioxide levels. Carbon dioxide present in the soil solution forms carbonic acid. This is a weak acid that solubilizes phosphorus compounds making them available to the plant. Second, increasing soil biology increases the amount of beneficial mycorrhizal fungi that can “mine” phosphorus from the soil and make it available to the plant. Applying soluble carbon to soil and limiting soil salinity both help to build soil biology.
Complexing is the process whereby negatively charged anions, such as phosphorus, or positively charged mono-valent (one positive charge) cations, such as potassium, are protected from undesirable reactions by chemically protecting the nutrient ion. One highly successful manufacturing technology involves the introduction of specialized carbon compounds that satisfy the highly reactive phosphorus charge. This process enables phosphorus to move in soil and assimilate into the plant efficiently. This can increase the relative phosphorus efficiency by as much as 25 times. This technology is used in all Redox products containing phosphorus, including Rootex and diKaP.

There are many options on the market to consider for phosphorus inputs. They can generally be grouped into five categories: Mined Phosphate Materials, Phosphoric Acid Derived Salts, Carbon Complexed Phosphate, Organic Derived Phosphate, and Soil Phosphate Primers. These groups each have unique characteristics and will be briefly discussed here.
Domestic mineral deposits in Florida, North Carolina, Idaho, and Utah are the primary source for mineral phosphate in the US. Morocco and China are the global leaders in phosphate mining with the US ranking third for bulk mined phosphate. Rock phosphate can be found as a primary component of many dry organic fertilizers, as well as, as a stand-alone soil amendment. The availability of phosphate in these materials is generally very low and pH dependent. Cost per unit P is very cheap, but due to the low phosphate availability the application rate per acre is very high and costly. The majority of the mined rock phosphate is used in manufacturing phosphoric acid.
Phosphoric Acid is derived from reacting rock phosphate minerals with sulfuric acid to make phosphoric acid and calcium sulphate (gypsum). Phosphoric acid is then combined with other fertility inputs to build multi-nutrient fertility products. For example, manufacturing 10-34-0, a common N and P fertilizer, combines anhydrous ammonia and phosphoric acid. These materials are chemically defined as salts, (containing an anion and cation held together by an ionic bond). These products are economical for manufacturing and transport because of their high concentration of plant nutrients and thus have a slightly higher cost per unit P than mined materials. The application rate however is also high because when applied to soil the phosphate will react quickly with soil cations, such as calcium and zinc, rendering it unavailable for plant uptake. Plants have the ability to “mine” phosphate from soil through soil pH modifications in the rhizosphere, but this process is energetically costly.
During phosphate fertilizer manufacturing it is possible to introduce carbon complexing agents to the reaction creating phosphate materials that are complexed by carbon. Carbon can also be mixed with the final product to achieve partial complexing of phosphate and other nutrients. These products generally contain humic acid, fulvic acid, or amino acids and are brown in color. The advantage of combining carbon with phosphate materials has two benefits. It feeds the soil microorganisms thereby increasing nutrient availability and improving soil tilth. It also maintains phosphate availability in soil by satisfying the electrical charge of the phosphate and preventing undesirable tie-up. There are many products on the market that fall into this category. They are difficult to compare on paper as they differ in the degree to which the phosphate in complexed, the carbon containing compounds used, price, and amount used per application. The cost per unit P is substantially higher than the previous mentioned groups, however the application rate per acre is also substantially lower due to high efficiency of the complex phosphate.
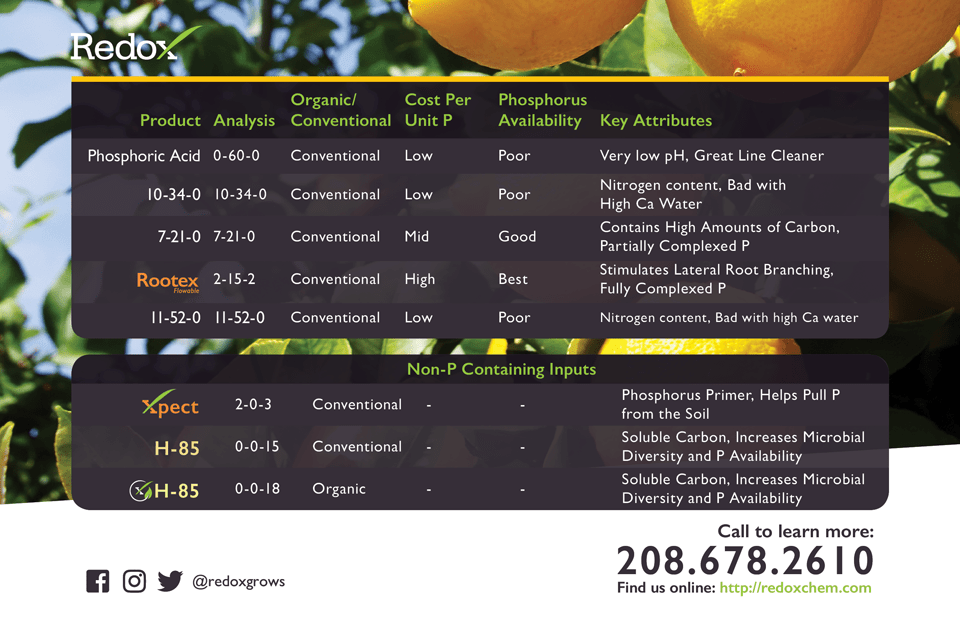
Many organic byproducts have high phosphate content, such as bone meal, blood meal, and livestock manures. Phosphate is bound within the molecular structure of the decomposing organic material. This means that P is not readily available and will require time to decompose to a form that is plant available. Different sources take different amounts of time to release the bound P. Many liquid organic products are allowed under organic regulations to use phosphoric acid as a pH modifier. This practice is common and provides some available phosphate.
Soils on the California Coast have what is considered high phosphate levels. This phosphate however is tightly bound in soil and not readily available to the plant. As discussed, earlier Soil Biology plays a large role in making phosphorus available. Additionally, a strong root system will be able to extract more phosphorus from the soil through root exudates production. Increasing soil biology and root strength through the application of soluble carbon and soil biostimulants will improve nutrient availability.
The Phosphorus market has a variety of product options ranging from simple mined and organic products that have been used for millennia to products born out of advanced manufacturing and formulating processes. Crops will respond favorably to improved phosphorus uptake and efficiency, enhancing the potential for improved crop returns.
Redox has several market leading phosphorus technologies. Rootex Flowable (2-15-2) is a high phosphorus fertilizer reacted with soluble carbon and L-amino acids. Redox complexing technology of phosphorus with soluble carbon chemistry reduces soil tie-up and significantly improves phosphorus efficiency. The six specific L-amino acids contained in Rootex Flowable increased soluble phosphorus, improve lateral root branching, root growth, and overall soil health. The unique formulation of Rootex Flowable is the new standard in phosphorus nutrition and root growth. H-85 is a multi-extraction humic and fulvic acid that contains short-, medium-, and long-chain soluble carbon molecules. H-85 is soluble across a broad pH range, improving soluble carbon availability in all soil types increasing soil microbial diversity, soil health, and nutrient availability.
Questions? Want to know more about Choose Your Phosphorus Wisely?
Subscribe to receive our Redox Bio-Nutrients updates